Origin: Extracted from minerals sylvite, carnallite, and potash. It may also be extracted from salt water and can be manufactured by crystallization from solution, flotation or electrostatic separation from suitable minerals.
It may also be generated in a laboratory by treating potassium hydroxide (or other potassium bases) with hydrochloric acid. Potassium may also be allowed to burn in the presence of chlorine gas to obtain this compound.
Also Known As:
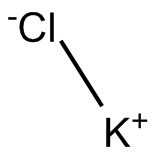
KCl
Potassium salt
Overview: Potassium chloride is an odorless white crystal composed of potassium and chlorine. It is highly soluble in water and it has a salt-like taste. It is used in medicine, scientific applications, and in food processing.
Its primary function is as a dietary supplement to help treat low levels of potassium in the blood. It can also be used as a salt substitute.
Common Dosage: 1500 mg per day







