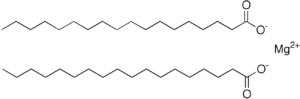
Origin: Produced by the reaction of sodium stearate with magnesium salts or by treating magnesium oxide with stearic acid.
Also Known As:
Magnesium octadecanoate
Overview: Magnesium stearate is a white, water-insoluble powder consisting of salt containing two equivalents of stearate (the anion of stearic acid) and one magnesium cation (Mg2+). Its applications exploit its softness, insolubility in many solvents, and low toxicity. It is used as a release agent and as a component or lubricant in the production of pharmaceuticals and cosmetics.
Common Dosage: Below 2500 mg per kg of body weight per day







